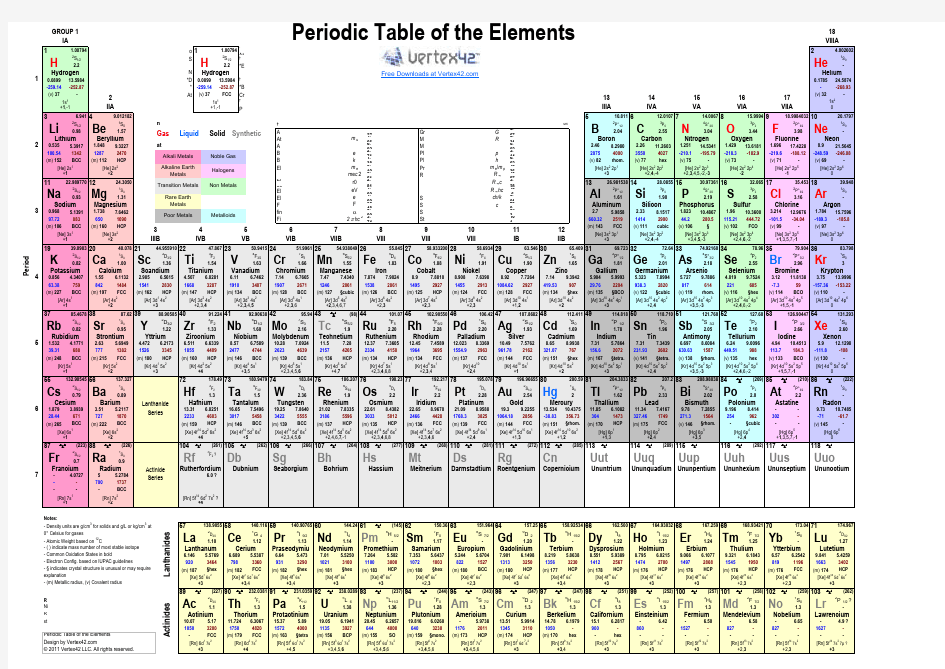
excel版的元素周期表periodic-table,超强大
Atomic Number
?Atomic Weight Symbol
?Ground-State Level *Electronegativity (Pauling)
Name
*Density [Note]
?Ionization Energy (eV)*Melting Point (°C)
*Boiling Point (°C)Atomic radius (pm)[Note]
Crystal Structure [Note]
?
Electron Configuration
Possible Oxidation States [Note]
Phase at STP
?
Common Constants
Source: https://www.360docs.net/doc/a93740311.html,
Absolute Zero -273.15 °C
Gravitation Constant 6.67428x10-11 m 3 kg -1 s -2
Atomic Mass Unit 1.660539x10-27 kg Molar Gas Constant
8.314472 J mol -1 K -1Categories
Avogadro Constant
6.022142x1023 mol -1Molar Volume (Ideal Gas)0.02241410 m 3/mol Base of Natural Logarithms 2.718281828PI
3.14159265358979Boltzmann constant 1.380650x10-23
J/K Planck Constant
6.626069x10-34 J s Electron Mass
9.10938215x10-31 kg Proton-Electron Mass Ratio 1836.152672470.5110 MeV Rydberg Constant
10 973 732 m -1Electron Radius (Classical) 2.8179403x10-15 m 3.289842x1015 Hz Electron Volt 1.602176x10-19 J 13.6057 eV Elementry Charge 1.602176x10-19 C Second Radiation Constant 0.01438769 m K Faraday Constant 96 485.3399 C/mol Speed of Light in a Vacuum 299 792 458 m/s fine-structure constant 0.0072973525
Speed of sound in air at STP 343.2 m/s [42]
First Radiation Constant
3.7417749x10-16
W m
2
Standard Pressure
101 325 Pa
{42}
References:
?https://www.360docs.net/doc/a93740311.html,, *https://www.360docs.net/doc/a93740311.html, (Mathematic),CRC Handbook of Chemistry and Physics 81st Edition, 2000-2001, and others
Bh
18Notes:
- Density units are g/cm 3 for solids and g/L or kg/cm 3 at 0°Celsius for gases
- Atomic Weight based on 12C
- ( ) indicate mass number of most stable isotope - Common Oxidation States in bold
- Electron Config. based on IUPAC guidelines
- § indicates crystal structure is unusual or may require explanation
- (m) Metallic radius, (v) Covalent radius ? 2011 Vertex42 LLC. All rights reserved.
Design by https://www.360docs.net/doc/a93740311.html,
Cn Periodic Table of the Elements Db
Sg
(v) 37
-
+1,-1
Li
GROUP 11
1.007942
4.002602
P e r i o d
1
1
1.00794
0.089913.5984He
1
S 0
2.2
2.2-
H
2
S 1/2H
2
S 1/2Hydrogen
Hydrogen
Helium
2IIA 0.089913.5984(v) 37
FCC +1,-1
-268.93-259.14-252.87-(v) 32
13IIIA 14IVA
15VA
16VIA
Free Downloads at https://www.360docs.net/doc/a93740311.html,
-1s 11s 11s 217VIIA
0.178524.5874
-259.14-252.870
9.012182
5
10.811
62
3
6.941
4
15.9994
9
18.9984032
10
12.0107
714.0067
820.1797
2
S 1/2Be
1
S 0B
G 2
P°1/2
C
3
P 0
N
3
P 2
F
3.04
3.442.04
2.554
S°3/2
O
2
P°3/2
Ne
1
S 0
0.98 1.57Gas Liquid Solid Synthetic
3.98
-
Lithium
Beryllium
m u R Boron
Carbon
Nitrogen
Oxygen
Fluorine
Neon
0.535 5.3917 1.8489.3227 2.468.2980 2.26p 207513.6181 1.69617.42280.911.2603 1.25114.5341 1.4294027-210.121.5645180.54134212872470Alkali Metals Noble Gas e -246.08(m) 152BCC
(m) 112
HCP
k h (v) 82-195.79-218.3rhom.
(v) 77hex
(v) 75-188.12-248.59-182.9-219.640003550 -
[He] 2s 1[He] 2s 2Alkaline Earth
Metals
Halogens m e m e /m p [He] 2s 2 2p 1
-
(v) 73[He] 2s 2 2p 2[He] 2s 2 2p 3[He] 2s 2 2p 4
[He] 2s 2 2p 5
-
(v) 69
-
(v) 71[He] 2s 2 2p 6
+1+2
me c 2R ∞+3+2,4,-4+2,3,4,5,-2,-3-2-10
3
11
22.989770
12
24.3050
Transition Metals Non Metals
r0R ∞c 18
39.948
15
30.97361
16
32.065
Na
2
S 1/2
Mg
1
S 0
17
35.453
13
26.981538
14
28.0855
Si
3
P 0
P
4
S°3/2
eV R ∞hc Al
2
P°1/2
S
3
P 2
Cl
2
P°3/2
2.58
3.16
Ar
1
S 0
0.93 1.31Rare Earth Metals
e ch/k 1.61
1.90
2.19
-
Sodium
Magnesium
F c
Aluminum
Silicon Phosphorus
Sulfur
Chlorine
Argon
0.968 5.1391 1.7387.6462Poor Metals Metalloids
a
2.710.4867 1.9610.3600
3.2145.9858 2.338.1517 1.82312.9676 1.78415.759697.7288365010902p hc 2660.32280.5115.2144
4.72-101.525191414290044.2-34.04-189.3-18
5.8(m) 186
BCC
(m) 160
HCP
3IIIB 4IVB 5VB 11IB 12IIB (m) 1436VIB 7VIIB 8VIII 9VIII §
(v) 102FCO
(v) 99
FCC
(v) 111cubic
(v) 106
-
(v) 97
-
[Ne] 3s 1[Ne] 3s 2[Ne] 3s 2 3p 1
[Ne] 3s 2 3p 2[Ne] 3s 2 3p 3[Ne] 3s 2 3p 4[Ne] 3s 2 3p 5[Ne] 3s 2 3p 6
+1+2
+3+2,4,-4+3,4,5,-3+2,4,6,-2+1,3,5,7,-10
10VIII 4
19
39.0983
20
63.3875984240.078
K
2
S 1/2
23
50.9415
24
51.996121
44.955910
22
47.86727
58.933200
28
58.693425
54.938049
26
55.84531
69.723
32
72.6429
63.546
30
65.40935
79.904
36
83.798
33
74.92160
34
78.96Ti
3
F 2V
4
F 3/2Ca
1
S 0
Sc
2
D 3/2Fe
5
D 4Co
1.83Cr
7
S 3Mn
6
S 5/24
F 9/2Ni
3
F 4Cu
1.88 1.912
S 1/2Zn
1
S 0Ga
1.90 1.652
P°1/2Ge
3
P 0As
1.81
2.014
S°3/2Se
3
P 2Br
2.18
2.552
P°3/2Kr
1
S 00.82 1.00 1.36
1.54 1.63 1.66 1.55
2.96
3
Potassium
Calcium
Scandium
Titanium
Vanadium
Chromium
Manganese
Iron
4.507 6.8281Gallium
Germanium
Arsenic
Selenium
Cobalt
Nickel
Copper
Zinc
0.856 4.3407 1.55 6.1132 2.985 6.5615 6.11 6.74627.14 6.7665Bromine
Krypton
8.97.88108.9087.63987.477.43407.8747.9024 5.904 5.9993 5.3237.89948.927.72647.149.3942 3.1211.8138 3.7513.99965.7279.7886 4.8199.7524328719103407190714841541283016682861149529271455267112462061153890729.762204938.329131084.622927419.53685-7.359-157.362820817614221-153.22(m) 227
BCC
(m) 197FCC
(m) 162HCP
(m) 147HCP
(m) 134§cubic
(m) 126BCC
(m) 125
BCC
(m) 128BCC
(m) 127
FCC
(m) 134§hex
(m) 135
HCP
(m) 124FCC
(m) 128rhom.
(v) 116
§hex
(v) 114§BCO
(v) 122
§cubic
(v) 119
BCO
(v) 110
-
[Ar] 4s 1[Ar] 4s 2[Ar] 3d 1 4s 2
[Ar] 3d 2 4s 2[Ar] 3d 3 4s 2[Ar] 3d 5 4s 1[Ar] 3d 5 4s 2+2,3,6+2,3,4,6,7
[Ar] 3d 10 4s 2
[Ar] 3d 10 4s 2 4p 1
[Ar] 3d 10 4s 2 4p 2
[Ar] 3d 10 4s 2 4p 3
[Ar] 3d 6 4s 2
[Ar] 3d 7 4s 2
[Ar] 3d 8 4s 2
[Ar] 3d 10 4s 1
+2,3+1,2[Ar] 3d 10 4s 2 4p 4
[Ar] 3d 10 4s 2 4p 5
[Ar] 3d 10 4s 2 4p 6
+1+2+3
+2,3,4+2,3,4,50
5
37
85.4678
3887.62
39
88.90585
+2+340
91.22441
92.90638
+2,4,6,-2+1,5,-1+2,4+3,5,-3+2,3
+2,344
101.0745
102.90550
4295.94
43(98)
48
112.41149
114.818
46
106.4247
107.8682
52
127.6053
126.90447
50
118.71051
121.760
54
131.293Rb
2
S 1/2
Sr
1
S 0
Y
2
D 3/2Zr
3
F 2Nb
6
D 1/2Mo
7
S 31.60
2.16Tc
6
S 5/2Ru
5
F 51.9
2.20Rh
4
F 9/2Pd
1
S 02.28
2.20Ag
2
S 1/2Cd
1
S 01.93
1.69In
2
P°1/2Sn
3
P 01.78
1.96Sb
4
S°3/2Te
3
P 22.05
2.10I
2
P°3/2Xe
1
S 02.66
2.60Rubidium
Strontium
Yttrium
Zirconium
0.820.95 1.22
1.336.511 6.6339Indium
Tin
Antimony
Tellurium
Rhodium
Palladium
Silver
Cadmium
1.532 4.1771
2.63 5.6949 4.472 6.21738.57 6.758910.287.0924Iodine
Xenon
Niobium
Molybdenum
Technetium
Ruthenium
12.457.458912.0238.336911.57.2812.377.36057.31 5.78647.317.343910.497.57628.658.9938 4.9410.4513 5.912.12986.6978.6084 6.249.0096152633451855440939.31688777138221574265233441502477474426234639961.782162321.07767196436951554.92963-111.8-108630.631587449.51988(m) 248
BCC
(m) 215FCC
113.7184.3156.62072231.932602(m) 146
BCC
(m) 139
BCC
(m) 180HCP
(m) 160HCP
(m) 134
FCC
(m) 137FCC
(m) 136HCP
(m) 134
HCP
(m) 167§tetra.
(v) 141
§tetra.
(m) 144
FCC
(m) 151
§hex
(v) 133
BCO
(v) 130
-(v) 138
§rhom.
(v) 135
hex
[Kr] 4d 4 5s 1
[Kr] 4d 5 5s 1[Kr] 4d 5 5s 2
[Kr] 4d 7 5s 1[Kr] 5s 1[Kr] 5s 2[Kr] 4d 1 5s 2
[Kr] 4d 2 5s 2
[Kr] 4d 10 5s 2 5p 1
[Kr] 4d 10 5s 2 5p 2
[Kr] 4d 10 5s 2 5p 3
[Kr] 4d 10 5s 2 5p 4
[Kr] 4d 8 5s 1[Kr] 4d 10[Kr] 4d 10 5s 1
[Kr] 4d 10 5s 2
[Kr] 4d 10 5s 2 5p 5
[Kr] 4d 10 5s 2 5p 6
+1+2+3+4+3,5+2,3,4,5,6+4,7+2,3,4,6,872
178.49
+3+2,4+3,5,-3+2,4,6,-2+2,3,4+2,4+1+26
55
132.90545
56
137.327
Lanthanide Series
73
180.9479
74
183.84
+1,5,7,-10
77
192.217
78
195.078
75
186.207
76
190.23
81
204.3833
82
207.2
79
196.96655
80
200.59
85
(210)
86
(222)
83
208.98038
84
(209)
Hf
3
F 2
Ta
4
F 3/2
Cs
2
S 1/2
Ba
1
S 0
Os
5
D 4
Ir
4
F 9/2
W
5
D 0
Re
6
S 5/2
Pt
3
D 3
Au
2
S 1/2
2.28 2.54Hg
1
S 0
Tl
2
P°1/2
2
1.62
Pb
3
P 0
Bi
4
S°3/2
2.33 2.02
Po
3
P 2
At
2
P°3/2
2.0 2.2Rn
1
S 0
0.790.89 1.3 1.5
2.36 1.9
2.2
2.2
-
Cesium
Barium
Hafnium
Tantalum
Tungsten
Rhenium
Osmium
Iridium
Platinum
Bismuth
Polonium
Astatine
Radon
Gold
Mercury
Thallium
Lead
13.31 6.825116.657.54961.879 3.8939 3.51 5.211722.618.438222.658.967019.257.864021.027.833513.53410.437511.85 6.108221.098.958819.39.22559.1968.414--11.347.41679.787.28559.7310.748528.446717271870223346033017545830335012246644283422555531865596-38.83356.7330414731768.338251064.182856254962302-327.461749271.31564-71-61.7(m) 265BCC
(m) 222BCC
(m) 159HCP
(m) 146BCC
(m) 135
HCP
(m) 136FCC
(m) 139BCC
(m) 137HCP
(m) 151
§rhom.
(m) 170HCP
(m) 139FCC
(m) 144FCC
-
§cubic
-
-
(m) 175FCC
(v) 146
§rhom.
(v) 145
-
[Xe] 6s 1[Xe] 6s 2[Xe] 4f 14 5d 2 6s 2
[Xe] 4f 14 5d 3 6s 2
[Xe] 4f 14 5d 4 6s 2
[Xe] 4f 14 5d 5 6s 2[Xe] 4f 14 5d 6 6s 2
[Xe] 4f 14 5d 7 6s 2
[Hg] 6p 2[Hg] 6p 3[Hg] 6p 4[Hg] 6p 5[Xe] 4f 14 5d 9 6s 1
[Xe] 4f 14 5d 10 6s 1
[Xe] 4f 14 5d 10 6s 2
[Hg] 6p 1[Hg] 6p 6
+1+2
+4
+5+2,3,4,5,6+2,4,6,7,-1+2,3,4,6,8+2,3,4,6+2,4+3,5
+2,4+1,3,5,7,-1
+1,3+1,2+1,3+2,47
87(223)
88Fr
2
S 1/2
Francium
(226)
Actinide Series
104
(261)
Ra
1
S 0
Rf
3
F 2 ?
Radium
Rutherfordium
107
(264)
108
(277)
105
(262)
106
(266)
111
(272)
112
(285)
109
(268)
110
(281)
118
115116(292)
Hs
Mt
117
113114(289)
Uuh Uus
Uut
Uuq Uup
Ds
Rg
Dubnium
Seaborgium
Bohrium
Hassium
Uuo
0.7
0.9Ununtrium Ununquadium Ununpentium
Ununhexium
Meitnerium Darmstadtium Roentgenium
Copernicium
Ununseptium
Ununoctium
- 4.07275 5.2784 6.0 ?
--7001737-
--
BCC
[Rn] 7s 1[Rn] 7s 2[Rn] 5f 14 6d 2 7s 2 ?
+1
+2+4
59
140.90765
60
144.2461
(145)
57
138.9055
La
2
D 3/2
1.10
Lanthanum
62
150.3663
151.964
A c t i n i d e s
89
58
140.116
Cerium L a n t h a n i d e s
66
162.50067
164.93032
64
157.25
65
158.92534
70
173.04
71
174.967
68
167.25969
168.93421
Ce
1
G°4Pr
4
I°9/21.12
1.13
Nd 5
I 4Pm
6
H°5/21.14
-
Sm
7
F 0Eu
8
S°7/21.17
-
Gd
9
D°2Tb
6
H°15/21.20
-
Dy
5
I 8Ho
4
I°15/21.22
1.23
Er
3
H 6Tm
2
F°7/21.24
1.25
Yb
1
S 0Lu
2
D 3/2
-
1.27
Thulium
Ytterbium
Europium
Gadolinium
Terbium
Dysprosium
5.4737.01 5.52507.264Holmium
Erbium
Praseodymium Neodymium
Promethium
Samarium
5.5827.353 5.6437 5.244Lutetium
6.146 5.5769 6.689 5.5387 6.64 5.86388.551 5.93898.7955.6704
7.901 6.1498
8.219 6.1843 6.57 6.2542
9.8416.02159.066 6.10779.321 5.42599203464798336093132901021310011001527131332501356300010721803822119616632700149728681545§hex
(m) 181§hex
(m) 18319508193230141225671474HCP
(m) 180§hex
(m) 1803402(m) 187§hex
(m) 182FCC
(m) 182HCP
(m) 178HCP
(m) 176BCC
(m) 180HCP
(m) 177HCP
(m) 176FCC
(m) 174HCP
(m) 176HCP
(m) 176HCP
[Xe] 5d 1
6s 2
[Xe] 4f 1 5d 1
6s 2
[Xe] 4f 3
6s 2
[Xe] 4f 4
6s 2
[Xe] 4f 5
6s 2
[Xe] 4f 6
6s 2
[Xe] 4f 7
6s 2
[Xe] 4f 7 5d 1
6s 2
[Xe] 4f 9
6s 2
+2,3+3[Xe] 4f 10
6s 2
[Xe] 4f 11
6s 2
[Xe] 4f 12
6s 2
[Xe] 4f 13
6s 2
+3+3,4+3,4
+3
+3+2,3+3,4+3+3+3[Xe] 4f 14
6s 2
[Xe] 4f 14 5d 1
6s 2
+2,3+2,3+3
(227)
90
232.0381
91
231.0359
92
238.0289
95
(243)
96
(247)
93
(237)
94
(244)U
5
L°6101
(258)
102
(259)
99
(252)
100
(257)
Ac
2
D 3/2
Th
3
F 2
Pa
4
K 11/2
Np
6
L 11/2Pu
7
F 0103
(262)
97
(247)
98
(251)Fm
Bk
6
H°15/2Cf
5
I 8Am
8
S°7/2Cm
9
D°21
S 0Lr
1.3
1.3
3
H 6Md
1.3
1.28
1.3
1.3
1.3
2
F°7/2No
1.3
Es
1.3
4
I°15/2Plutonium
Americium
Curium
Berkelium
2
P°1/2 ?1.1
1.3
1.5
1.38
1.36
Californium
Einsteinium
Fermium
Mendelevium
-
Actinium
Thorium Protactinium
Uranium
Neptunium
Nobelium Lawrencium
10.07 5.1711.724 6.306715.37 5.8919.05 6.1941- 5.9738
13.51 5.991420.45 6.265719.816 6.026011353927- 6.58
- 6.65
- 6.42
- 6.50
10503200175048201572400064440006403230- 4.9 ?
14.78 6.197915.1 6.28171050-900-1176201113453110827-827-860-1527-1627--FCC (m) 179FCC
(m) 163§tetra
(m) 156BCP
(m) 173HCP
(m) 174HCP
(m) 155SO
(m) 159§mono.
[Rn] 5f 7
7s 2[Rn] 5f 7 6d 7s
2
--------[Rn] 6d 1
7s 2
[Rn] 6d 2
7s 2
[Rn] 5f 2
6d 1 7s 2
[Rn] 5f 3 6d 1
7s 2
[Rn] 5f 4 6d 1
7s 2
[Rn] 5f 6 7s 2[Rn] 5f 9
7s 2
[Rn] 5f 10
7s 2
[Rn] 5f 11 7s
2
[Rn] 5f 12 7s
2
--(m) 170hex
-hex
[Rn] 5f 13 7s 2
[Rn] 5f 14 7s 2
[Rn] 5f 14 7s 2
7p ?
+3+4+4,5
+3,4,5,6+3,4,5,6+3,4,5,6+3,4,5,6+3+2,3
+2,3
+3
+3,4+3
+3
+3
